Lyophilization Services for Biopharmaceuticals Market Scope: Expanding Opportunities and Growing Demand
The Lyophilization Services for Biopharmaceuticals Market is experiencing significant growth, fueled by the increasing demand for biologic drugs and the need for specialized preservation techniques to ensure their stability and shelf life. Lyophilization, or freeze-drying, is a critical process for maintaining the potency of temperature-sensitive biologics such as vaccines, monoclonal antibodies, peptides, and gene therapies. This process removes water from a formulation, ensuring that the biological product remains stable during storage and transportation without compromising its efficacy. With the growing prevalence of chronic diseases, the rise in biologics-based treatments, and the expansion of the vaccine sector, the demand for lyophilization services is expected to continue rising in the coming years.
Get the Sample PDF @ https://www.theinsightpartners.com/reports/lyophilization-services-for-biopharmaceuticals-market
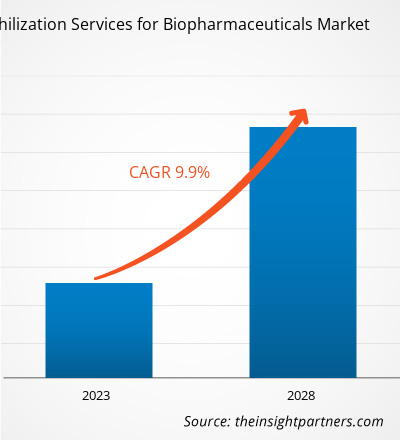
The Lyophilization Services for Biopharmaceuticals Market Scope has broadened considerably due to the expansion of biologic drugs and the shift toward outsourcing pharmaceutical manufacturing to contract development and manufacturing organizations (CDMOs). Biopharmaceutical companies are increasingly turning to CDMOs for their lyophilization needs due to the latter’s specialized expertise, scalable operations, and regulatory compliance capabilities. The lyophilization services for biopharmaceuticals market is expected to grow from US$ 2,051.41 million in 2022 to US$ 3,586.55 million by 2028; it is estimated to register a CAGR of 9.9% from 2023 to 2028.
As the biopharmaceutical sector grows, so does the need for technologies that ensure the stability of biologic products. The global rise in chronic diseases and the development of biologic treatments for conditions such as cancer, autoimmune disorders, and genetic disorders has further driven the demand for lyophilization services. These products require sophisticated stabilization methods due to their sensitivity to heat and moisture, making lyophilization an essential process for their preservation. Additionally, the growing focus on vaccines, particularly in light of global health challenges like the COVID-19 pandemic, has highlighted the importance of maintaining product stability for large-scale distribution.
The Lyophilization Services for Biopharmaceuticals Market is further strengthened by technological advancements in freeze-drying equipment and process optimization. Modern lyophilization equipment incorporates sophisticated automation systems, enhanced temperature control, and energy-efficient processes, allowing for greater reliability and consistency in the freeze-drying process. Additionally, the integration of real-time data analytics, process monitoring, and digital systems has significantly improved process optimization, enabling better product quality, reduced cycle times, and more efficient production. These advancements are making lyophilization more cost-effective and are helping service providers meet the growing demand for high-quality biologics.
Another factor driving market expansion is the trend of increasing pharmaceutical manufacturing outsourcing. Contract development and manufacturing organizations (CDMOs) are becoming vital players in the biopharmaceutical landscape by providing specialized services like lyophilization for biologic drugs. These organizations not only offer technical expertise and advanced equipment but also provide scalability, which is crucial for accommodating both large-scale and niche market demands. By outsourcing lyophilization processes, pharmaceutical companies can reduce capital expenditures, streamline their operations, and improve their overall time-to-market for new therapies.
The Lyophilization Services for Biopharmaceuticals Market is experiencing significant growth, fueled by the increasing demand for biologic drugs and the need for specialized preservation techniques to ensure their stability and shelf life. Lyophilization, or freeze-drying, is a critical process for maintaining the potency of temperature-sensitive biologics such as vaccines, monoclonal antibodies, peptides, and gene therapies. This process removes water from a formulation, ensuring that the biological product remains stable during storage and transportation without compromising its efficacy. With the growing prevalence of chronic diseases, the rise in biologics-based treatments, and the expansion of the vaccine sector, the demand for lyophilization services is expected to continue rising in the coming years.
Get the Sample PDF @ https://www.theinsightpartners.com/reports/lyophilization-services-for-biopharmaceuticals-market
The Lyophilization Services for Biopharmaceuticals Market Scope has broadened considerably due to the expansion of biologic drugs and the shift toward outsourcing pharmaceutical manufacturing to contract development and manufacturing organizations (CDMOs). Biopharmaceutical companies are increasingly turning to CDMOs for their lyophilization needs due to the latter’s specialized expertise, scalable operations, and regulatory compliance capabilities. The lyophilization services for biopharmaceuticals market is expected to grow from US$ 2,051.41 million in 2022 to US$ 3,586.55 million by 2028; it is estimated to register a CAGR of 9.9% from 2023 to 2028.
As the biopharmaceutical sector grows, so does the need for technologies that ensure the stability of biologic products. The global rise in chronic diseases and the development of biologic treatments for conditions such as cancer, autoimmune disorders, and genetic disorders has further driven the demand for lyophilization services. These products require sophisticated stabilization methods due to their sensitivity to heat and moisture, making lyophilization an essential process for their preservation. Additionally, the growing focus on vaccines, particularly in light of global health challenges like the COVID-19 pandemic, has highlighted the importance of maintaining product stability for large-scale distribution.
The Lyophilization Services for Biopharmaceuticals Market is further strengthened by technological advancements in freeze-drying equipment and process optimization. Modern lyophilization equipment incorporates sophisticated automation systems, enhanced temperature control, and energy-efficient processes, allowing for greater reliability and consistency in the freeze-drying process. Additionally, the integration of real-time data analytics, process monitoring, and digital systems has significantly improved process optimization, enabling better product quality, reduced cycle times, and more efficient production. These advancements are making lyophilization more cost-effective and are helping service providers meet the growing demand for high-quality biologics.
Another factor driving market expansion is the trend of increasing pharmaceutical manufacturing outsourcing. Contract development and manufacturing organizations (CDMOs) are becoming vital players in the biopharmaceutical landscape by providing specialized services like lyophilization for biologic drugs. These organizations not only offer technical expertise and advanced equipment but also provide scalability, which is crucial for accommodating both large-scale and niche market demands. By outsourcing lyophilization processes, pharmaceutical companies can reduce capital expenditures, streamline their operations, and improve their overall time-to-market for new therapies.
Lyophilization Services for Biopharmaceuticals Market Scope: Expanding Opportunities and Growing Demand
The Lyophilization Services for Biopharmaceuticals Market is experiencing significant growth, fueled by the increasing demand for biologic drugs and the need for specialized preservation techniques to ensure their stability and shelf life. Lyophilization, or freeze-drying, is a critical process for maintaining the potency of temperature-sensitive biologics such as vaccines, monoclonal antibodies, peptides, and gene therapies. This process removes water from a formulation, ensuring that the biological product remains stable during storage and transportation without compromising its efficacy. With the growing prevalence of chronic diseases, the rise in biologics-based treatments, and the expansion of the vaccine sector, the demand for lyophilization services is expected to continue rising in the coming years.
Get the Sample PDF @ https://www.theinsightpartners.com/reports/lyophilization-services-for-biopharmaceuticals-market
The Lyophilization Services for Biopharmaceuticals Market Scope has broadened considerably due to the expansion of biologic drugs and the shift toward outsourcing pharmaceutical manufacturing to contract development and manufacturing organizations (CDMOs). Biopharmaceutical companies are increasingly turning to CDMOs for their lyophilization needs due to the latter’s specialized expertise, scalable operations, and regulatory compliance capabilities. The lyophilization services for biopharmaceuticals market is expected to grow from US$ 2,051.41 million in 2022 to US$ 3,586.55 million by 2028; it is estimated to register a CAGR of 9.9% from 2023 to 2028.
As the biopharmaceutical sector grows, so does the need for technologies that ensure the stability of biologic products. The global rise in chronic diseases and the development of biologic treatments for conditions such as cancer, autoimmune disorders, and genetic disorders has further driven the demand for lyophilization services. These products require sophisticated stabilization methods due to their sensitivity to heat and moisture, making lyophilization an essential process for their preservation. Additionally, the growing focus on vaccines, particularly in light of global health challenges like the COVID-19 pandemic, has highlighted the importance of maintaining product stability for large-scale distribution.
The Lyophilization Services for Biopharmaceuticals Market is further strengthened by technological advancements in freeze-drying equipment and process optimization. Modern lyophilization equipment incorporates sophisticated automation systems, enhanced temperature control, and energy-efficient processes, allowing for greater reliability and consistency in the freeze-drying process. Additionally, the integration of real-time data analytics, process monitoring, and digital systems has significantly improved process optimization, enabling better product quality, reduced cycle times, and more efficient production. These advancements are making lyophilization more cost-effective and are helping service providers meet the growing demand for high-quality biologics.
Another factor driving market expansion is the trend of increasing pharmaceutical manufacturing outsourcing. Contract development and manufacturing organizations (CDMOs) are becoming vital players in the biopharmaceutical landscape by providing specialized services like lyophilization for biologic drugs. These organizations not only offer technical expertise and advanced equipment but also provide scalability, which is crucial for accommodating both large-scale and niche market demands. By outsourcing lyophilization processes, pharmaceutical companies can reduce capital expenditures, streamline their operations, and improve their overall time-to-market for new therapies.
0 Yorumlar
0 hisse senetleri
421 Views
0 önizleme